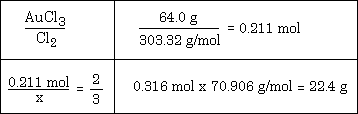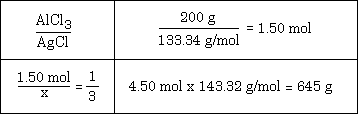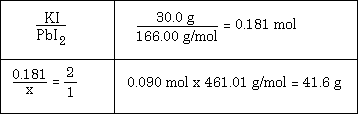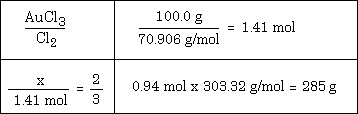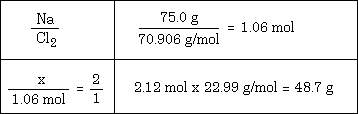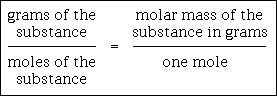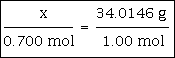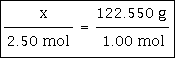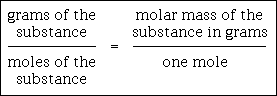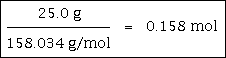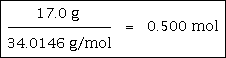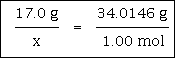Stoichiometry
The Meaning of the Word
The word stoichiometry derives from two Greek words: stoicheion (meaning "element") and metron (meaning "measure"). Stoichiometry deals with calculations about the masses (sometimes volumes) of reactants and products involved in a chemical reaction. It is a very mathematical part of chemistry, so be prepared for lots of calculator use.
Jeremias Benjaim Richter (1762-1807) was the first to lay down the principles of stoichiometry. In 1792 he wrote:
"Die stöchyometrie (Stöchyometria) ist die Wissenschaft die quantitativen oder Massenverhältnisse zu messen, in welchen die chymischen Elemente gegen einander stehen."
[Stoichiometry is the science of measuring the quantitative proportions or mass ratios in which chemical elements stand to one another.]
What You Should Expect
The most common stoichiometric problem will present you with a certain amount of a reactant and then ask how much of a product can be formed. Here is a generic chemical equation:
2 A + 2B ---> 3C
Here is a typically-worded problem (often called a "mass to mass" problem):
Given 20.0 grams of A and sufficient B, how many grams of C can be produced? You will need to use molar ratios, molar masses, balancing and interpreting equations, and conversions between grams and moles. If you struggled with those in class, welcome to the club. Go back and review them if you need to, because if you can't do that stuff, you can't do stoichiometry.
The Steps Involved in Solving Mass-Mass Stoichiometry Problems
Molar Ratios
The molar ratio will assume a place of central importance in solving stoichiometry problems. The sources for these ratios are the coefficients of a balanced equation. We will look at what a molar ratio is and then a brief word on how to recognize which ratio to use in a problem.
2 H2 + O2 ---> 2 H2O
What is the molar ratio between H2 and O2?
Answer: two to one. 2:1 or 2/1
It's recommended that you explicitly write a one in the denominator of the ratio.
What is the molar ratio between O2 and H2O?
Answer: one to two. As a fraction, it is: 1/2
What is the molar ratio between H2 and H2O?
Answer: two to two or: 2/2
As you well know, this reduces to one to one. However, we will use the unreduced fraction in the lessons to follow.
Here's another example equation:
2 O3 ---> 3 O2 The exact molar ratio you would use depends on how the problem is worded.
What is the molar ratio between O3 and O2?
What is the molar ratio between O2 and O3?
The first one, of course, is: 2/3
And the second simply reverses the numbers: 3/2
Before looking at the practice problems, an important point: the coefficients of a reaction only give the ratio in which substances react. They do not in any way tell you HOW MUCH is reacting.
The word stoichiometry derives from two Greek words: stoicheion (meaning "element") and metron (meaning "measure"). Stoichiometry deals with calculations about the masses (sometimes volumes) of reactants and products involved in a chemical reaction. It is a very mathematical part of chemistry, so be prepared for lots of calculator use.
Jeremias Benjaim Richter (1762-1807) was the first to lay down the principles of stoichiometry. In 1792 he wrote:
"Die stöchyometrie (Stöchyometria) ist die Wissenschaft die quantitativen oder Massenverhältnisse zu messen, in welchen die chymischen Elemente gegen einander stehen."
[Stoichiometry is the science of measuring the quantitative proportions or mass ratios in which chemical elements stand to one another.]
What You Should Expect
The most common stoichiometric problem will present you with a certain amount of a reactant and then ask how much of a product can be formed. Here is a generic chemical equation:
2 A + 2B ---> 3C
Here is a typically-worded problem (often called a "mass to mass" problem):
Given 20.0 grams of A and sufficient B, how many grams of C can be produced? You will need to use molar ratios, molar masses, balancing and interpreting equations, and conversions between grams and moles. If you struggled with those in class, welcome to the club. Go back and review them if you need to, because if you can't do that stuff, you can't do stoichiometry.
The Steps Involved in Solving Mass-Mass Stoichiometry Problems
- Make sure the chemical equation is correctly balanced.
- Using the molar mass of the given substance, convert the mass given in the problem to moles.
- Construct a molar proportion (two molar ratios set equal to each other) following the guidelines which will be set out in the tutorial. Use it to convert to moles of the unknown.
- Using the molar mass of the unkown substance, convert the moles just calculated to mass.
Molar Ratios
The molar ratio will assume a place of central importance in solving stoichiometry problems. The sources for these ratios are the coefficients of a balanced equation. We will look at what a molar ratio is and then a brief word on how to recognize which ratio to use in a problem.
2 H2 + O2 ---> 2 H2O
What is the molar ratio between H2 and O2?
Answer: two to one. 2:1 or 2/1
It's recommended that you explicitly write a one in the denominator of the ratio.
What is the molar ratio between O2 and H2O?
Answer: one to two. As a fraction, it is: 1/2
What is the molar ratio between H2 and H2O?
Answer: two to two or: 2/2
As you well know, this reduces to one to one. However, we will use the unreduced fraction in the lessons to follow.
Here's another example equation:
2 O3 ---> 3 O2 The exact molar ratio you would use depends on how the problem is worded.
What is the molar ratio between O3 and O2?
What is the molar ratio between O2 and O3?
The first one, of course, is: 2/3
And the second simply reverses the numbers: 3/2
Before looking at the practice problems, an important point: the coefficients of a reaction only give the ratio in which substances react. They do not in any way tell you HOW MUCH is reacting.
| molar ratio problems | |
| File Size: | 27 kb |
| File Type: | docx |
Mole to Mole
The solution procedure used below involves making two ratios and setting them equal to each other. This is called a proportion. One ratio will come from the coefficients of the balanced equation and the other will be constructed from the problem. The ratio set up from data in the problem will almost always be the one with an unknown in it.
You will then cross-multiply and divide to get the answer.
What happens if the equation isn't balanced? Answer - balance it. You cannot do these problems correctly without a balanced equation.
How will I know which substances to use in the ratio? Answer - you will have to read the problem and understand the words in it.
Here is the first equation we'll use:
N2 + 3 H2 ---> 2 NH3 Problem
#1: if we have 2.00 mol of N2 reacting with sufficient H2, how many moles of NH3 will be produced?
Solution Comments
That means the ratio from the equation is: 2/1
The ratio from the data in the problem will be: x/2.00
The proportion (setting the two ratios equal) is: x/2.00 = 2/1
Solving by cross-multiplying gives x = 4.00 mol of NH3 produced.
Example #2 - Suppose 6.00 mol of H2 reacted with sufficient nitrogen. How many moles of ammonia would be produced?
Let's use this ratio to set up the proportion: NH3-H2
That means the ratio from the equation is: 2/3
The ratio from the data in the problem will be: x/6.00
The proportion (setting the two ratios equal) is: x/6.00 = 2/3
Solving by cross-multiplying and dividing gives x = 4.00 mol of NH3 produced.
Example #3 - We want to produce 2.75 mol of NH3. How many moles of nitrogen would be required?
Before the solution, a brief comment: notice that hydrogen IS NOT mentioned in this problem. If any substance ISN'T mentioned in the problem, then assume there is a sufficient quantity of it on hand. Since that substance isn't part of the problem, then it's not part of the solution.
Let's use this ratio to set up the proportion: NH3-N2
That means the ratio from the equation is: 2/1
The ratio from the data in the problem will be: x/2.75
The proportion (setting the two ratios equal) is: x/2.75 = 2/1
Solving by cross-multiplying and dividing (plus rounding off to three significant figures) gives x = 1.38 mol of N2 needed.
The solution procedure used below involves making two ratios and setting them equal to each other. This is called a proportion. One ratio will come from the coefficients of the balanced equation and the other will be constructed from the problem. The ratio set up from data in the problem will almost always be the one with an unknown in it.
You will then cross-multiply and divide to get the answer.
What happens if the equation isn't balanced? Answer - balance it. You cannot do these problems correctly without a balanced equation.
How will I know which substances to use in the ratio? Answer - you will have to read the problem and understand the words in it.
Here is the first equation we'll use:
N2 + 3 H2 ---> 2 NH3 Problem
#1: if we have 2.00 mol of N2 reacting with sufficient H2, how many moles of NH3 will be produced?
Solution Comments
- The ratio from the problem will have N2 and NH3 in it.
- How do you know which number goes on top or bottom in the ratios? Answer: it does not matter, except that you observe the next point ALL THE TIME.
- When making the two ratios, be 100% certain that numbers are in the same relative positions. For example, if the value associated with NH3 is in the numerator, then MAKE SURE it is in both numerators.
- Use the coefficients of the two substances to make the ratio from the equation.
- Why isn't H2 involved in the problem? Answer: The word "sufficient" removes it from consideration.
That means the ratio from the equation is: 2/1
The ratio from the data in the problem will be: x/2.00
The proportion (setting the two ratios equal) is: x/2.00 = 2/1
Solving by cross-multiplying gives x = 4.00 mol of NH3 produced.
Example #2 - Suppose 6.00 mol of H2 reacted with sufficient nitrogen. How many moles of ammonia would be produced?
Let's use this ratio to set up the proportion: NH3-H2
That means the ratio from the equation is: 2/3
The ratio from the data in the problem will be: x/6.00
The proportion (setting the two ratios equal) is: x/6.00 = 2/3
Solving by cross-multiplying and dividing gives x = 4.00 mol of NH3 produced.
Example #3 - We want to produce 2.75 mol of NH3. How many moles of nitrogen would be required?
Before the solution, a brief comment: notice that hydrogen IS NOT mentioned in this problem. If any substance ISN'T mentioned in the problem, then assume there is a sufficient quantity of it on hand. Since that substance isn't part of the problem, then it's not part of the solution.
Let's use this ratio to set up the proportion: NH3-N2
That means the ratio from the equation is: 2/1
The ratio from the data in the problem will be: x/2.75
The proportion (setting the two ratios equal) is: x/2.75 = 2/1
Solving by cross-multiplying and dividing (plus rounding off to three significant figures) gives x = 1.38 mol of N2 needed.
| Mole to mole (practice) | |
| File Size: | 26 kb |
| File Type: | docx |
Mole to Mass
The solution procedure used below involves making two ratios and setting them equal to each other. This is called a proportion. One ratio will come from the coefficients of the balanced equation and the other will be constructed from the problem. The ratio set up from data in the problem will almost always be the one with an unknown in it.
You will then cross-multiply and divide to get the answer.
However, there is one addition to the above technique. One of the values will need to be expressed in moles. This could be either a reactant or a product. In either case, moles will have to be converted to grams or the reverse.
Suppose you are given a mass in the problem. You will need to convert this to moles FIRST. You do this by dividing the mass given by the molar mass of the substances.
Suppose you are asked for a mass as an answer. You will convert the moles you calculated in the proportion to grams. You do this by multiplying the moles by the molar mass of the substance.
Here is the first equation we'll use:
2 KClO3 ---> 2 KCl + 3 O2 Problem #1: 1.50 mol of KClO3 decomposes. How many grams of O2 will be produced?
Let's use this ratio to set up the proportion: KClO3/O2
That means the ratio from the equation is: 2/3
The ratio from the data in the problem will be: 1.50/x
The proportion (setting the two ratios equal) is: 1.50/x = 2/3
Cross-multiplying and dividing gives x = 2.25 mol of O2 produced.
2.25 mol x 32.0 g/mol = 72.0 grams. The 32.0 g/mol is the molar mass of O2.
Example #2 - If 80.0 grams of O2 was produced, how many moles of KClO3 decomposed?
Let's use this ratio to set up the proportion: O2/KClO3
That means the ratio from the equation is: 3/2
The ratio from the data in the problem will be: 2.50/x
The 2.50 mole came from 80.0 g ÷ 32.0 g/mol. The 32.0 g/mol is the molar mass of O2. Be careful to keep in mind that oxygen is O2, not just O.
The proportion (setting the two ratios equal) is: 2.50/x = 3/2
Solving by cross-multiplying and dividing gives x = 1.67 mol of KClO3 decomposed.
Example #3 - We want to produce 2.75 mol of KCl. How many grams of KClO3 would be required?
Let's use this ratio to set up the proportion: KCl/KClO3
That means the ratio from the equation is: 2/2
The ratio from the data in the problem will be: 2.75/x
The proportion (setting the two ratios equal) is: 2.75/x = 2/2
Hopefully, it's pretty easy to see that 2.75 mol of KClO3 are needed. However, the question wants grams for an answer.
2.75 mol times 122.55 g/mol = 337 grams completes the task. The 122.55 g/mol is the molar mass of KClO3.
The solution procedure used below involves making two ratios and setting them equal to each other. This is called a proportion. One ratio will come from the coefficients of the balanced equation and the other will be constructed from the problem. The ratio set up from data in the problem will almost always be the one with an unknown in it.
You will then cross-multiply and divide to get the answer.
However, there is one addition to the above technique. One of the values will need to be expressed in moles. This could be either a reactant or a product. In either case, moles will have to be converted to grams or the reverse.
Suppose you are given a mass in the problem. You will need to convert this to moles FIRST. You do this by dividing the mass given by the molar mass of the substances.
Suppose you are asked for a mass as an answer. You will convert the moles you calculated in the proportion to grams. You do this by multiplying the moles by the molar mass of the substance.
Here is the first equation we'll use:
2 KClO3 ---> 2 KCl + 3 O2 Problem #1: 1.50 mol of KClO3 decomposes. How many grams of O2 will be produced?
Let's use this ratio to set up the proportion: KClO3/O2
That means the ratio from the equation is: 2/3
The ratio from the data in the problem will be: 1.50/x
The proportion (setting the two ratios equal) is: 1.50/x = 2/3
Cross-multiplying and dividing gives x = 2.25 mol of O2 produced.
2.25 mol x 32.0 g/mol = 72.0 grams. The 32.0 g/mol is the molar mass of O2.
Example #2 - If 80.0 grams of O2 was produced, how many moles of KClO3 decomposed?
Let's use this ratio to set up the proportion: O2/KClO3
That means the ratio from the equation is: 3/2
The ratio from the data in the problem will be: 2.50/x
The 2.50 mole came from 80.0 g ÷ 32.0 g/mol. The 32.0 g/mol is the molar mass of O2. Be careful to keep in mind that oxygen is O2, not just O.
The proportion (setting the two ratios equal) is: 2.50/x = 3/2
Solving by cross-multiplying and dividing gives x = 1.67 mol of KClO3 decomposed.
Example #3 - We want to produce 2.75 mol of KCl. How many grams of KClO3 would be required?
Let's use this ratio to set up the proportion: KCl/KClO3
That means the ratio from the equation is: 2/2
The ratio from the data in the problem will be: 2.75/x
The proportion (setting the two ratios equal) is: 2.75/x = 2/2
Hopefully, it's pretty easy to see that 2.75 mol of KClO3 are needed. However, the question wants grams for an answer.
2.75 mol times 122.55 g/mol = 337 grams completes the task. The 122.55 g/mol is the molar mass of KClO3.
| Mole to Mass (practice) | |
| File Size: | 26 kb |
| File Type: | docx |
Mass to Mass
This is the most common type of stoichiometric problem in high school.
There are four steps involved in solving these problems:
Comments
STOP!!! Go back to the start of this and re-read it. Notice that I give four steps (and some advice) in how to solve the example problems just below. My advice is to keep going back to those steps as you examine the samples below.
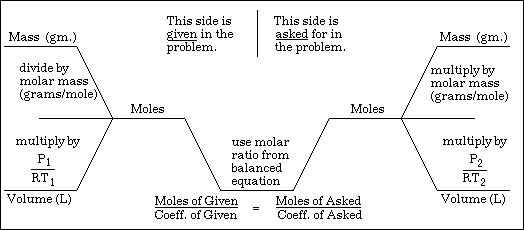
Here is a picture of the steps involved in solving mass-mass problems.
This is the most common type of stoichiometric problem in high school.
There are four steps involved in solving these problems:
- Make sure you are working with a properly balanced equation.
- Convert grams of the substance given in the problem to moles.
- Construct two ratios - one from the problem and one from the equation and set them equal. Solve for "x," which is usually found in the ratio from the problem.
- Convert moles of the substances just solved for into grams.
Comments
- Double check the equation.
- DON'T use the same molar mass in steps two and four. Your teacher is aware of this and, on a multiple choice test, will provide the answer arrived at by making this mistake. You have been warned!
- Don't multiply the molar mass of a substance by the coefficient in the problem BEFORE using it in one of the steps above. For example, if the formula says 2 H2O, DON'T use 36.0 g/mol, use 18.0 g/mol.
- Don't round off until the very last answer. In other words, don't clear your calculator after step two and write down a value of 3 or 4 significant figures to use in the next step. Round off only once after all calculations are done.
STOP!!! Go back to the start of this and re-read it. Notice that I give four steps (and some advice) in how to solve the example problems just below. My advice is to keep going back to those steps as you examine the samples below.
Here is a picture of the steps involved in solving mass-mass problems.
As you can see, the bottom portion includes mass-volume problems. These type problems are not discussed in this website.
Each of the example problems below has an associated image which lays out the solution. Reading from left to right, the top row gives:
1. the molar ratio used in the problem's solution.
2. the conversion of the grams given in the problem to moles.
The second row gives:
3. the molar proportion used to convert from moles of the given to moles of the unknown.
4. the conversion of moles of the unknown back to grams.
Each of the example problems below has an associated image which lays out the solution. Reading from left to right, the top row gives:
1. the molar ratio used in the problem's solution.
2. the conversion of the grams given in the problem to moles.
The second row gives:
3. the molar proportion used to convert from moles of the given to moles of the unknown.
4. the conversion of moles of the unknown back to grams.
Example #1 - How many grams of chlorine can be liberated from the decomposition of 64.0 g. of AuCl3 by this reaction: 2 AuCl3 ---> 2 Au + 3 Cl2
**One question often asked is "Where did the value of 303.32 come from?" Answer - it's the molar mass of AuCl3. Keep this answer in mind as you wonder about where other numbers come from in a given solution.
You might also want to consider looking at the solution to the problem and try to fit it to the list of steps given above.
Example #2 - Calculate the mass of AgCl that can be prepared from 200. g of AlCl3 and sufficient AgNO3, using this equation: 3 AgNO3 + AlCl3 --> 3 AgCl + Al(NO3)3
You might also want to consider looking at the solution to the problem and try to fit it to the list of steps given above.
Example #2 - Calculate the mass of AgCl that can be prepared from 200. g of AlCl3 and sufficient AgNO3, using this equation: 3 AgNO3 + AlCl3 --> 3 AgCl + Al(NO3)3
Example #3 - Given this equation: 2 KI + Pb(NO3)2 --> PbI2 + 2 KNO3 calculate mass of PbI2 produced by reacting of 30.0 g KI with excess Pb(NO3)2
Example #4 - How many grams of AuCl3 can be made from 100.0 grams of chlorine by this reaction: 2 Au + 3 Cl2 ---> 2 AuCl3
Example #5 - How many grams of Na are required to react completely with 75.0 grams of chlorine using this reaction: 2 Na + Cl2 ---> 2 NaCl
| work sheet (practice) | |
| File Size: | 54 kb |
| File Type: | docx |
Moles to Grams
In chemistry, the mole is the standard measurement of amount. When substances react, they do so in simple ratios of moles. However, balances give readings in grams. Balances DO NOT give readings in moles.
So the problem is that, when we compare amounts of one substance to another using moles, we must convert from grams, since this is the information we get from balances.
There are three steps to converting moles of a substance to grams:
The three steps above can be expressed in the following proportion:
In chemistry, the mole is the standard measurement of amount. When substances react, they do so in simple ratios of moles. However, balances give readings in grams. Balances DO NOT give readings in moles.
So the problem is that, when we compare amounts of one substance to another using moles, we must convert from grams, since this is the information we get from balances.
There are three steps to converting moles of a substance to grams:
- Determine how many moles are given in the problem.
- Calculate the molar mass of the substance.
- Multiply step one by step two.
The three steps above can be expressed in the following proportion:
This proportion is a symbolic equation. When you solve a particular problem, you insert the proper numbers & units into the proper places of the symbolic equation and then you solve using cross-multiplication and division. Also, do not attach units to the unknown. Let it be simply the letter "x." The proper unit should evolve naturally from solving the proportion and cancellation of units.
Make sure you have a periodic table and a calculator handy.
Example #1 - calculate how many grams are in 0.700 moles of H2O2
Step One: The problem will tell you how many moles are present. Look for the word "mole" or the unit "mol." The number immediately proceeding it will be how many moles.
I suppose that a problem can be worded in such a way that the number of moles comes after the unit, but that type of trickery isn't very common in high school.
0.700 moles are given in the problem.
Step Two: You need to know the molar mass of the substance. Please refer to the lessons about calculating the molecular weight and molar mass of a substance if you are not sure how to calculate a molar mass.
The molar mass of H2O2 is 34.0146 grams/mole. You may wish to pause and calculate this value, if you desire the practice.
Step Three: You multiply the moles given by the substance's molar mass:
0.700 mole x 34.0146 grams/mole = 23.8 grams The answer of 23.8 g has been rounded to three significant figures because the 0.700 value had the least number of significant figures in the problem.
If this problem were set up like the proportion above, you would have this:
Make sure you have a periodic table and a calculator handy.
Example #1 - calculate how many grams are in 0.700 moles of H2O2
Step One: The problem will tell you how many moles are present. Look for the word "mole" or the unit "mol." The number immediately proceeding it will be how many moles.
I suppose that a problem can be worded in such a way that the number of moles comes after the unit, but that type of trickery isn't very common in high school.
0.700 moles are given in the problem.
Step Two: You need to know the molar mass of the substance. Please refer to the lessons about calculating the molecular weight and molar mass of a substance if you are not sure how to calculate a molar mass.
The molar mass of H2O2 is 34.0146 grams/mole. You may wish to pause and calculate this value, if you desire the practice.
Step Three: You multiply the moles given by the substance's molar mass:
0.700 mole x 34.0146 grams/mole = 23.8 grams The answer of 23.8 g has been rounded to three significant figures because the 0.700 value had the least number of significant figures in the problem.
If this problem were set up like the proportion above, you would have this:
Then, cross-multiply and divide to solve for the unknown.
Example #2 - convert 2.50 moles of KClO3 to grams.
2.50 moles is given in the problem.
The molar mass for KClO3 is 122.550 grams/mole. Please note the unit of 'grams/mole.' It is important for proper cancelling of units that you remember to write this unit down when using a molar mass.
Following step three, we obtain:
2.50 moles x 122.550 grams/mole = 306.375 grams The answer should be rounded off to three significant figures, resulting in 306 g. as the correct answer. Note how the mole in the numerator and the mole in the denominator cancel.
If this problem were set up like the proportion above, you would have this:
Example #2 - convert 2.50 moles of KClO3 to grams.
2.50 moles is given in the problem.
The molar mass for KClO3 is 122.550 grams/mole. Please note the unit of 'grams/mole.' It is important for proper cancelling of units that you remember to write this unit down when using a molar mass.
Following step three, we obtain:
2.50 moles x 122.550 grams/mole = 306.375 grams The answer should be rounded off to three significant figures, resulting in 306 g. as the correct answer. Note how the mole in the numerator and the mole in the denominator cancel.
If this problem were set up like the proportion above, you would have this:
| Moles to grams (practice) | |
| File Size: | 25 kb |
| File Type: | docx |
Grams to Moles
In chemistry, the mole is the standard measurement of amount. However, balances DO NOT give readings in moles. Balances give readings in grams.
So the problem is that, while we compare amounts of one substance to another using moles, we must also use grams, since this is the information we get from balances.
There are three steps to converting grams of a substance to moles.
The three steps above can be expressed in the following proportion:
In chemistry, the mole is the standard measurement of amount. However, balances DO NOT give readings in moles. Balances give readings in grams.
So the problem is that, while we compare amounts of one substance to another using moles, we must also use grams, since this is the information we get from balances.
There are three steps to converting grams of a substance to moles.
- Determine how many grams are given in the problem.
- Calculate the molar mass of the substance.
- Divide step one by step two.
The three steps above can be expressed in the following proportion:
This proportion is a symbolic equation. When you solve a particular problem, you insert the proper numbers & units into the proper places of the symbolic equation and then you solve using cross-multiplication and division. Also, do not attach units to the unknown. Let it be simply the letter "x." The proper unit should evolve naturally from solving the proportion and cancellation of units.
Make sure you have a periodic table and a calculator handy.
Example #1 - Convert 25.0 grams of KMnO4 to moles.
Step One: The problem will tell you how many grams are present. Look for the unit of grams. The number immediately proceeding it will be how many grams. Common abbreviations for grams include g (just the letter).
The problem gives us 25.0 grams.
Step Two: You need to know the molar mass of the substance. Please refer to the lessons about calculating the molecular weight and molar mass of a substance if you are not sure how to calculate a molar mass.
The molar mass of KMnO4 is 158.034 grams/mole. Please take a moment and calculate the molar mass of KMnO4, just to be sure.
Step Three: You divide the grams given by the substance's molar mass:
Make sure you have a periodic table and a calculator handy.
Example #1 - Convert 25.0 grams of KMnO4 to moles.
Step One: The problem will tell you how many grams are present. Look for the unit of grams. The number immediately proceeding it will be how many grams. Common abbreviations for grams include g (just the letter).
The problem gives us 25.0 grams.
Step Two: You need to know the molar mass of the substance. Please refer to the lessons about calculating the molecular weight and molar mass of a substance if you are not sure how to calculate a molar mass.
The molar mass of KMnO4 is 158.034 grams/mole. Please take a moment and calculate the molar mass of KMnO4, just to be sure.
Step Three: You divide the grams given by the substance's molar mass:
Then, cross-multiply and divide to solve for the unknown.
Example #2 - calculate how many moles are in 17.0 grams of H2O2
17.0 grams are given in the text of the problem. The molar mass is 34.0146 grams/mole, which is calculated using the formula and the atomic weights on a periodic table. You may want to calculate this value as a double-check of your mastery of calculating molar masses.
Example #2 - calculate how many moles are in 17.0 grams of H2O2
17.0 grams are given in the text of the problem. The molar mass is 34.0146 grams/mole, which is calculated using the formula and the atomic weights on a periodic table. You may want to calculate this value as a double-check of your mastery of calculating molar masses.
This answer has been rounded to three significant figures because of the 17.0.
This problem can also be set up as a proportion:
This problem can also be set up as a proportion:
| Grams to Moles (practice) | |
| File Size: | 33 kb |
| File Type: | docx |