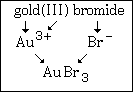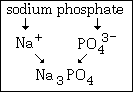Nomenclature
| Table of Common Ions (A MUST HAVE!) | |
| File Size: | 372 kb |
| File Type: | |
A binary compound is one made of two different elements. There can be one of each element such as in NaCl or KF. There can also be several of each element such as Na2O or AlBr3.
Please remember that all elements involved in this lesson have ONLY ONE charge. That includes BOTH the cation AND the anion involved in the formula.
Points to remember about naming a compound from its formula
Step #1 - Look at first element and name it. Result of this step = hydrogen.
Step #2 - Look at second element. Use root of its full name ( which is sulf-) plus the ending "-ide." Result of this step = sulfide.
These two steps give the full name of H2S. Notice that the presence of the subscript is ignored. There are other types of binary compounds where you must pay attention to the subscript. Those compounds involve cations with variable charges. Your teacher will tell you which ones you will be held responsible for.
Example 2: Write the name of the following formula: NaCl
Step #1 - Look at first element and name it. Result of this step = sodium.
Step #2 - Look at second element. Use root of its full name ( which is chlor-) plus the ending "-ide." Result of this step = chloride.
Example 3: Write the name of the following formula: MgBr2
Step #1 - Look at first element and name it. Result of this step = magnesium.
Step #2 - Look at second element. Use root of its full name ( which is brom-) plus the ending "-ide." Result of this step = bromide.
Note the presence of the subscript does not play a role in this name.
Example 4: Write the name of the following formula: KCl
The first part of the name comes from the first element symbol: potassium. The second part of the name comes from the root of the second symbol plus '-ide,' therefore chlor + ide = chloride.
This compound is named potassium chloride
Example 5: Write the name of the following formula: Na2S
First symbol is Na, so the first part of the name is sodium. (Note the presence of the subscript does not play a role in this name.) Second element is sulfur (from the symbol S), so the name is sulf + ide = sulfide.
This compound is named sodium sulfide.
Three possible mistakes to be aware of:
1) Often students forget to use the suffix "-ide." For example, BaS is named "barium sulfide." An unaware student might want to name it "barium sulfur."
2) Make sure that the second name is the root plus "-ide." An unaware student might want to name BaS as "barium sulfuride." NaBr is not named sodium bromineide, the corect answer is sodium bromide.
3) There is a set of binary compounds which are named using Roman numerals. Students often confuse the two sets of rules. For example, a student might want to name Na2S as sodium (I) sulfide. While it is never wrong to use the Roman numerals, your teacher will probably want you to only use Roman numerals on certain cations.
Here are examples of common roots:
Cl: chlor-
F: fluor-
Br: brom-
O: ox-
I: iod-
N: nitr-
Please remember that all elements involved in this lesson have ONLY ONE charge. That includes BOTH the cation AND the anion involved in the formula.
Points to remember about naming a compound from its formula
- The order for names in a binary compound is first the cation, then the anion.
- Use the name of cation with a fixed oxidation state directly from the periodic table.
- The name of the anion will be made from the root of the element's name plus the suffix "-ide."
Step #1 - Look at first element and name it. Result of this step = hydrogen.
Step #2 - Look at second element. Use root of its full name ( which is sulf-) plus the ending "-ide." Result of this step = sulfide.
These two steps give the full name of H2S. Notice that the presence of the subscript is ignored. There are other types of binary compounds where you must pay attention to the subscript. Those compounds involve cations with variable charges. Your teacher will tell you which ones you will be held responsible for.
Example 2: Write the name of the following formula: NaCl
Step #1 - Look at first element and name it. Result of this step = sodium.
Step #2 - Look at second element. Use root of its full name ( which is chlor-) plus the ending "-ide." Result of this step = chloride.
Example 3: Write the name of the following formula: MgBr2
Step #1 - Look at first element and name it. Result of this step = magnesium.
Step #2 - Look at second element. Use root of its full name ( which is brom-) plus the ending "-ide." Result of this step = bromide.
Note the presence of the subscript does not play a role in this name.
Example 4: Write the name of the following formula: KCl
The first part of the name comes from the first element symbol: potassium. The second part of the name comes from the root of the second symbol plus '-ide,' therefore chlor + ide = chloride.
This compound is named potassium chloride
Example 5: Write the name of the following formula: Na2S
First symbol is Na, so the first part of the name is sodium. (Note the presence of the subscript does not play a role in this name.) Second element is sulfur (from the symbol S), so the name is sulf + ide = sulfide.
This compound is named sodium sulfide.
Three possible mistakes to be aware of:
1) Often students forget to use the suffix "-ide." For example, BaS is named "barium sulfide." An unaware student might want to name it "barium sulfur."
2) Make sure that the second name is the root plus "-ide." An unaware student might want to name BaS as "barium sulfuride." NaBr is not named sodium bromineide, the corect answer is sodium bromide.
3) There is a set of binary compounds which are named using Roman numerals. Students often confuse the two sets of rules. For example, a student might want to name Na2S as sodium (I) sulfide. While it is never wrong to use the Roman numerals, your teacher will probably want you to only use Roman numerals on certain cations.
Here are examples of common roots:
Cl: chlor-
F: fluor-
Br: brom-
O: ox-
I: iod-
N: nitr-
| given formula, write name (pracitce problems) | |
| File Size: | 15 kb |
| File Type: | docx |
Points to remember about writing the formula from the name
This file, Charge-Crossing.html, shows a technique, slightly different from below, to figure out formulas.
This file, Least-Common-Multiple.html, shows still a different slant on how to figure out a formula.
Example 1: Write the formula from the following name: sodium bromide
Step #1 - Write down the symbol and charge of the first word. Result = Na+
Step #2 - Write down the symbol and charge of the second word. Result = Br¯
Step #3 - Use the minimum number of cations and anions needed to make the sum of all charges in the formula equal zero. In this case, only one Na+ and one Br¯ are required.
The resulting formula is NaBr.
Example 2: Write the formula from the following name: potassium chloride
Step #1 - Write down the symbol and charge of the first word. Result = K+
Step #2 - Write down the symbol and charge of the second word. Result = Cl¯
Step #3 - Use the minimum number of cations and anions needed to make the sum of all charges in the formula equal zero. In this case, only one K+ and one Cl¯ are required.
The resulting formula is KCl.
Example 3: Write the formula from the following name: barium iodide
Step #1 - Write down the symbol and charge of the first word. Result = Ba2+
Step #2 - Write down the symbol and charge of the second word. Result = I¯
Step #3 - Use the minimum number of cations and anions needed to make the sum of all charges in the formula equal zero. In this case, only one Ba2+ is required, but two I¯ are required.
Why? Answer - Two negative one charges are required because there is one postive two charge. Only in this way can the total charge of the formula be zero.
The resulting formula is BaI2.
Example 4: Write the formula from the following name: aluminum chloride
Step #1 - Write down the symbol and charge of the first word. Result = Al3+
Step #2 - Write down the symbol and charge of the second word. Result = Cl¯
Step #3 - Use the minimum number of cations and anions needed to make the sum of all charges in the formula equal zero. In this case, only one Al3+ is required, but three Cl¯ are required.
Why? Answer - Three negative one charges are required because there is one positive three charge. Only in this way can the total charge of the formula be zero.
The resulting formula is AlCl3.
Example 5: Write the name of the following formula: magnesium oxide
Step #1 - Write down the symbol and charge of the first word. Result = Mg2+
Step #2 - Write down the symbol and charge of the second word. Result = O2¯
Step #3 - Use the minimum number of cations and anions needed to make the sum of all charges in the formula equal zero. In this case, one Mg2+ is required, as well as one O2¯.
Why? Answer - One positive two charge is counterbalanced by one negative two charge. This gives a zero total charge for the formula.
The resulting formula is MgO.
Example 6: Write the name of the following formula: aluminum oxide
Step #1 - Write down the symbol and charge of the first word. Result = Al3+
Step #2 - Write down the symbol and charge of the second word. Result = O2¯
Step #3 - Use the minimum number of cations and anions needed to make the sum of all charges in the formula equal zero. In this case, two Al3+ are required and three O2¯.
Why? Answer - This is the only possible way to get the positive and negative charges equal and keep the numbers to a minimum. Note that the positive charge is a +6 and the negative charge is a -6.
Also, keep in mind that you cannot change the charges to make a formula correct.
The resulting formula is Al2O3.
Warning: beware of the temptation to write the above formula as Al3O2.
- The order in a formula is first the cation, then the anion.
- You must know the charges associated with each cation and anion.
- The sum of the positive charge and the sum of the negative charges MUST add up to zero.
- You MAY NOT adjust the charges of the cations or anions to get a total charge of zero.
- You MAY adjust the subscripts to get a total charge of zero.
This file, Charge-Crossing.html, shows a technique, slightly different from below, to figure out formulas.
This file, Least-Common-Multiple.html, shows still a different slant on how to figure out a formula.
Example 1: Write the formula from the following name: sodium bromide
Step #1 - Write down the symbol and charge of the first word. Result = Na+
Step #2 - Write down the symbol and charge of the second word. Result = Br¯
Step #3 - Use the minimum number of cations and anions needed to make the sum of all charges in the formula equal zero. In this case, only one Na+ and one Br¯ are required.
The resulting formula is NaBr.
Example 2: Write the formula from the following name: potassium chloride
Step #1 - Write down the symbol and charge of the first word. Result = K+
Step #2 - Write down the symbol and charge of the second word. Result = Cl¯
Step #3 - Use the minimum number of cations and anions needed to make the sum of all charges in the formula equal zero. In this case, only one K+ and one Cl¯ are required.
The resulting formula is KCl.
Example 3: Write the formula from the following name: barium iodide
Step #1 - Write down the symbol and charge of the first word. Result = Ba2+
Step #2 - Write down the symbol and charge of the second word. Result = I¯
Step #3 - Use the minimum number of cations and anions needed to make the sum of all charges in the formula equal zero. In this case, only one Ba2+ is required, but two I¯ are required.
Why? Answer - Two negative one charges are required because there is one postive two charge. Only in this way can the total charge of the formula be zero.
The resulting formula is BaI2.
Example 4: Write the formula from the following name: aluminum chloride
Step #1 - Write down the symbol and charge of the first word. Result = Al3+
Step #2 - Write down the symbol and charge of the second word. Result = Cl¯
Step #3 - Use the minimum number of cations and anions needed to make the sum of all charges in the formula equal zero. In this case, only one Al3+ is required, but three Cl¯ are required.
Why? Answer - Three negative one charges are required because there is one positive three charge. Only in this way can the total charge of the formula be zero.
The resulting formula is AlCl3.
Example 5: Write the name of the following formula: magnesium oxide
Step #1 - Write down the symbol and charge of the first word. Result = Mg2+
Step #2 - Write down the symbol and charge of the second word. Result = O2¯
Step #3 - Use the minimum number of cations and anions needed to make the sum of all charges in the formula equal zero. In this case, one Mg2+ is required, as well as one O2¯.
Why? Answer - One positive two charge is counterbalanced by one negative two charge. This gives a zero total charge for the formula.
The resulting formula is MgO.
Example 6: Write the name of the following formula: aluminum oxide
Step #1 - Write down the symbol and charge of the first word. Result = Al3+
Step #2 - Write down the symbol and charge of the second word. Result = O2¯
Step #3 - Use the minimum number of cations and anions needed to make the sum of all charges in the formula equal zero. In this case, two Al3+ are required and three O2¯.
Why? Answer - This is the only possible way to get the positive and negative charges equal and keep the numbers to a minimum. Note that the positive charge is a +6 and the negative charge is a -6.
Also, keep in mind that you cannot change the charges to make a formula correct.
The resulting formula is Al2O3.
Warning: beware of the temptation to write the above formula as Al3O2.
| Given name, write formula (practice problems) | |
| File Size: | 15 kb |
| File Type: | docx |
A binary compound is one made of two different elements. There can be one of each element such as in CuCl or FeO. There can also be several of each element such as Fe2O3 or SnBr4.
This lesson shows you how to name binary compounds from the formula when a cation of variable charge is involved. The four formulas above are all examples of this type.
The cations involved in this lesson have AT LEAST TWO charges. The anions involved have only one charge.
The type of naming you will learn about is called the Stock system or Stock's system. It was designed by Alfred Stock (1876-1946), a German chemist and first published in 1919. In his own words, he considered the system to be "simple, clear, immediately intelligible, capable of the most general application."
In 1924, a German commission recommended Stock's system be adopted with some changes. For example, FeCl2,which would have been named iron(2)-chloride according to Stock's original idea, became iron(II) chloride in the revised proposal. In 1934, Stock approved of the Roman numerals, but felt it better to keep the hyphen and drop the parenthesis. This suggestion has not been followed, but the Stock system remains in use world-wide.
Example #1: Write the name for: FeCl2
Step #1 - the first part of the name is the unchanged name of the first element in the formula. In this example, it would be iron.
Step #2 - the result from step one WILL be followed by a Roman numeral. Here is how to determine its value:
Step #3 - the anion is named in the usual manner of stem plus "ide."
The correct name of the example is iron(II) chloride.
Example #2: name this compound: CuCl2
In this example, I've explained it differently. Compare it to the one above. Example #4 is also explained this way.
Example #3: Write the name for: Fe2O3
Step #1 - the first part of the name is the unchanged name of the first element in the formula. In this example, it would be iron.
Step #2 - the result from step one WILL be followed by a Roman numeral. Here is how to determine its value:
Step #3 - the anion is named in the usual manner of stem plus "ide."
The correct name of the example is iron(III) oxide.
Example #4: name this compound: SnO
This lesson shows you how to name binary compounds from the formula when a cation of variable charge is involved. The four formulas above are all examples of this type.
The cations involved in this lesson have AT LEAST TWO charges. The anions involved have only one charge.
The type of naming you will learn about is called the Stock system or Stock's system. It was designed by Alfred Stock (1876-1946), a German chemist and first published in 1919. In his own words, he considered the system to be "simple, clear, immediately intelligible, capable of the most general application."
In 1924, a German commission recommended Stock's system be adopted with some changes. For example, FeCl2,which would have been named iron(2)-chloride according to Stock's original idea, became iron(II) chloride in the revised proposal. In 1934, Stock approved of the Roman numerals, but felt it better to keep the hyphen and drop the parenthesis. This suggestion has not been followed, but the Stock system remains in use world-wide.
Example #1: Write the name for: FeCl2
Step #1 - the first part of the name is the unchanged name of the first element in the formula. In this example, it would be iron.
Step #2 - the result from step one WILL be followed by a Roman numeral. Here is how to determine its value:
- multiply the charge of the anion (the Cl) by its subscript. Ignore the fact that it is negative. In this example it is one times two equals two.
- divide this result by the subscript of the cation (the Fe). This is the value of the Roman numeral to use. In this example, it is two divided by one equals two.
- The value of the Roman number equals the positive charge on the cation in this formula.
Step #3 - the anion is named in the usual manner of stem plus "ide."
The correct name of the example is iron(II) chloride.
Example #2: name this compound: CuCl2
In this example, I've explained it differently. Compare it to the one above. Example #4 is also explained this way.
- The first part of the name comes from the first element symbol: copper.
- The Roman numeral is II, because 2 chlorides equal -2, so the Cu must be +2. (It must be +2 so that the total charge equals zero.
- The second part of the name comes from the root of the second symbol plus 'ide,' therefore chlor + ide = chloride.
Example #3: Write the name for: Fe2O3
Step #1 - the first part of the name is the unchanged name of the first element in the formula. In this example, it would be iron.
Step #2 - the result from step one WILL be followed by a Roman numeral. Here is how to determine its value:
- multiply the charge of the anion (the O) by its subscript. Ignore the fact that it is negative. In this example, it is two times three equals six.
- divide this result by the subscript of the cation (the Fe). This is the value of the Roman numeral to use. In this example, it is six divided by two equals three.
- Note: this value of the Roman number equals the positive charge on the cation.
Step #3 - the anion is named in the usual manner of stem plus "ide."
The correct name of the example is iron(III) oxide.
Example #4: name this compound: SnO
- First symbol is Sn, so the first part of the name is tin.
- The Roman numeral is II, because one oxygen = -2, so the one tin equals +2.
- Second element is oxygen (from the symbol O), so the name is ox + ide = oxide.
| given formula, write name (practice) | |
| File Size: | 16 kb |
| File Type: | docx |
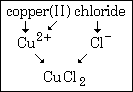
Example #1 - Write the formula for: copper(II) chloride
Step #1 - the first word tells you the symbol of the cation. In this case it is Cu.
Step #2 - the Roman numeral WILL tell you the charge on the cation. In this case it is a positive two.
Step #3 - the anion symbol and charge comes from the second name. In this case, chloride means Cl¯.
Step #4 - remembering the rule that a formula must have zero total charge, you write the formula CuCl2.
On writing the formula from the ions, you may want to review Charge-Crossing.html or Least-Common-Multiple.html for more information.
This graphic summarizes example #1:
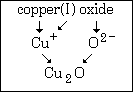
Example #2 - Write the formula for: copper(I) oxide
Step #1 - the first word tells you the symbol of the cation. It is Cu.
Step #2 - the Roman numeral WILL tell you the charge on the cation. It is a positive one.
Step #3 - the anion symbol and charge comes from the second name. Oxide means O2¯.
Step #4 - since a formula must have zero total charge, you write the formula Cu2O.
This graphic summarizes example #2:
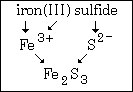
Example #3 - Write the formula for: iron(III) sulfide
Step #1 - the symbol of the cation is Fe.
Step #2 - the charge on the cation is a positive three. remember, that comes from the Roman numeral.
Step #3 - Sulfide (the anion) means S2¯.
Step #4 - since a formula must have zero total charge, you write the formula Fe2S3.
Look at Least-Common-Multiple.html if you're not sure about how the subscripts in iron(III) sulfide came to be.
This graphic summarizes example #3:
Step #1 - the symbol of the cation is Fe.
Step #2 - the charge on the cation is a positive three. remember, that comes from the Roman numeral.
Step #3 - Sulfide (the anion) means S2¯.
Step #4 - since a formula must have zero total charge, you write the formula Fe2S3.
Look at Least-Common-Multiple.html if you're not sure about how the subscripts in iron(III) sulfide came to be.
This graphic summarizes example #3:
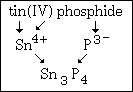
Example #4 - Write the formula for: tin(IV) phosphide First symbol is Sn from the name tin.
The Roman numeral IV gives +4 as tin's charge.
Phosphide give P3¯.
This compound's formula is Sn3P4.
This graphic summarizes example #4:
The Roman numeral IV gives +4 as tin's charge.
Phosphide give P3¯.
This compound's formula is Sn3P4.
This graphic summarizes example #4:
Here is an extra graphic for gold(III) bromide:
| given name, write formula (practice) | |
| File Size: | 16 kb |
| File Type: | docx |
A binary compound is one made of two different elements. There can be one of each element such as in CO or NO. There can also be several of each element such as BF3 or OCl2.
This lesson shows you how to name binary compounds from the formula when two nonmetals are involved. The four formulas above are all examples of this type. Important point to remember: NO metals (which act as the cation) are involved. That means one of the nonmetals will be acting in the positive role while the other is negative.
In fact, you do not even need to know the charges, since the formula comes right from the element names and their prefixes. Be aware that heavy use of Greek number prefixes are used in this lesson.Here are the first ten:
one: mono- six: hexa-
two: di- seven: hepta-
three: tri- eight: octa-
four: tetra- nine: nona-
five: penta- ten: deca-
Example #1 - write the name for N2O.
Example #2 - write the name for NO2.
Step #1 - part of the first name is the unchanged name of the first element in the formula. In the examples above, it would be nitrogen.
If the subscript of the first element is 2 or more, you add a prefix to the name. In the first example above, you would write dinitrogen. If the subscript is one as in the second example above, you DO NOT use a prefix. You simply write the name, in this example it would be nitrogen.
Step #2 - the anion is named in the usual manner of stem plus "ide." In addition, a prefix is added. In the first example, the prefix is "mono-" since there is one oxygen. In the second example, use "di-" because of two oxygens.
The correct names of the two examples are dinitrogen monoxide and nitrogen dioxide.
Note that "monoxide" is written rather than "monooxide." It sounds better when spoken out loud.
Example #3 - write the name for IF7.
Step #1 - the first element is iodine and there is only one. This part of the name will be "iodine", NOT "monoiodine."
Step #2 - the second element is fluorine, so "fluoride" is used. Since there are seven, the prefix "hepta" is used.
The name of this compound is iodine heptafluoride.
Example #4 - write the name for N2O5.
Step #1 - the first element is nitrogen and there are two. This part of the name will be "dinitrogen."
Step #2 - the second element is oxygen, so "oxide" is used. Since there are five, the prefix "penta" is used.
The name of this compound is "dinitrogen pentaoxide." Many write is as "dinitrogen pentoxide." The ChemTeam believes that both are considered correct, but the second is to be prefered.
Example #5 - write the name for XeF2.
The first part of the name comes from the first element's name: xenon. Since there is only one atom present, no prefix is used.
The second part of the name comes from the root of the second symbol plus 'ide' as well as the prefix "di-,"therefore di + fluor + ide = difluoride.
This compound is named xenon difluoride.
Example #6 - write the name for N2O4.
The first part of the name comes from the first element's name: nitrogen. Since there are two atoms, the prefix "di-" is used giving dinitrogen.
The second part of the name comes from the root of the second symbol plus 'ide' as well as the prefix "tetra-,"therefore tetr + ox + ide = tetroxide.
This compound is named dinitrogen tetroxide. Notice the dropping of the "a" in tetra.
Just a reminder: this system of naming does not really have an offically accepted name, but is often called the Greek system (or method). It involves use of Greek prefixes when naming binary compounds of two nonmetals.
Sometimes you will see the Stock system applied to these types of compounds. Here is what the IUPAC currently says about that practice: "The Stock notation can be applied to both cations and anions, but preferably should not be applied to compounds between nonmetals."
This lesson shows you how to name binary compounds from the formula when two nonmetals are involved. The four formulas above are all examples of this type. Important point to remember: NO metals (which act as the cation) are involved. That means one of the nonmetals will be acting in the positive role while the other is negative.
In fact, you do not even need to know the charges, since the formula comes right from the element names and their prefixes. Be aware that heavy use of Greek number prefixes are used in this lesson.Here are the first ten:
one: mono- six: hexa-
two: di- seven: hepta-
three: tri- eight: octa-
four: tetra- nine: nona-
five: penta- ten: deca-
Example #1 - write the name for N2O.
Example #2 - write the name for NO2.
Step #1 - part of the first name is the unchanged name of the first element in the formula. In the examples above, it would be nitrogen.
If the subscript of the first element is 2 or more, you add a prefix to the name. In the first example above, you would write dinitrogen. If the subscript is one as in the second example above, you DO NOT use a prefix. You simply write the name, in this example it would be nitrogen.
Step #2 - the anion is named in the usual manner of stem plus "ide." In addition, a prefix is added. In the first example, the prefix is "mono-" since there is one oxygen. In the second example, use "di-" because of two oxygens.
The correct names of the two examples are dinitrogen monoxide and nitrogen dioxide.
Note that "monoxide" is written rather than "monooxide." It sounds better when spoken out loud.
Example #3 - write the name for IF7.
Step #1 - the first element is iodine and there is only one. This part of the name will be "iodine", NOT "monoiodine."
Step #2 - the second element is fluorine, so "fluoride" is used. Since there are seven, the prefix "hepta" is used.
The name of this compound is iodine heptafluoride.
Example #4 - write the name for N2O5.
Step #1 - the first element is nitrogen and there are two. This part of the name will be "dinitrogen."
Step #2 - the second element is oxygen, so "oxide" is used. Since there are five, the prefix "penta" is used.
The name of this compound is "dinitrogen pentaoxide." Many write is as "dinitrogen pentoxide." The ChemTeam believes that both are considered correct, but the second is to be prefered.
Example #5 - write the name for XeF2.
The first part of the name comes from the first element's name: xenon. Since there is only one atom present, no prefix is used.
The second part of the name comes from the root of the second symbol plus 'ide' as well as the prefix "di-,"therefore di + fluor + ide = difluoride.
This compound is named xenon difluoride.
Example #6 - write the name for N2O4.
The first part of the name comes from the first element's name: nitrogen. Since there are two atoms, the prefix "di-" is used giving dinitrogen.
The second part of the name comes from the root of the second symbol plus 'ide' as well as the prefix "tetra-,"therefore tetr + ox + ide = tetroxide.
This compound is named dinitrogen tetroxide. Notice the dropping of the "a" in tetra.
Just a reminder: this system of naming does not really have an offically accepted name, but is often called the Greek system (or method). It involves use of Greek prefixes when naming binary compounds of two nonmetals.
Sometimes you will see the Stock system applied to these types of compounds. Here is what the IUPAC currently says about that practice: "The Stock notation can be applied to both cations and anions, but preferably should not be applied to compounds between nonmetals."
| given formula, write name (practice) | |
| File Size: | 16 kb |
| File Type: | docx |
Example #1 - write the formula for dinitrogen trioxide.
Example #2 - write the name for carbon monoxide.
Step #1 - the first name will tell you the first element in the formula. In the first example above, it would be N and in the second, C.
If there is a prefix on the name, this gives the subscript on the element. In the first example above, the "di-" tells you there are two nitrogens. Absence of a prefix, as in the second example, says there is only one of that element involved.
Step #2 - the anion name tells you the element; oxide means oxygen. Once again, the prefix will tell you how many of the element are involved. "Tri-" means three and "mono-" means one.
The correct formulas of the two examples are N2O3 and CO.
Note that "monoxide' is written rather than "oxide" when there is one atom of the second element involved. Note also that when one element of the first atom is involved, no "mono-" is used. Monocarbon monoxide is just as wrong as carbon oxide.
Example #3 - write the formula for bromine pentafluoride.
Step #1 - the first symbol is Br and its subscript will be a one, which is understoo to be present.
Step #2 - the second element is fluorine, so F is used. The prefix "penta-" indicates a subscript of 5.
The formula of this compound is BrF5.
Example #4 - write the formula for diphosphorous pentoxide.
Step #1 - the first symbol is P and the subscript is 2.
Step #2 - pentoxide says five oxygens are involved.
The formula of this compound is P2O5.
Example #5 - write the formula for iodine heptafluoride.
Step #1 - the first symbol is I and the subscript is 1. Again it is understood to be there.
Step #2 - heptafluoride says 7 florides are involved.
The formula of this compound is IF7.
Example #2 - write the name for carbon monoxide.
Step #1 - the first name will tell you the first element in the formula. In the first example above, it would be N and in the second, C.
If there is a prefix on the name, this gives the subscript on the element. In the first example above, the "di-" tells you there are two nitrogens. Absence of a prefix, as in the second example, says there is only one of that element involved.
Step #2 - the anion name tells you the element; oxide means oxygen. Once again, the prefix will tell you how many of the element are involved. "Tri-" means three and "mono-" means one.
The correct formulas of the two examples are N2O3 and CO.
Note that "monoxide' is written rather than "oxide" when there is one atom of the second element involved. Note also that when one element of the first atom is involved, no "mono-" is used. Monocarbon monoxide is just as wrong as carbon oxide.
Example #3 - write the formula for bromine pentafluoride.
Step #1 - the first symbol is Br and its subscript will be a one, which is understoo to be present.
Step #2 - the second element is fluorine, so F is used. The prefix "penta-" indicates a subscript of 5.
The formula of this compound is BrF5.
Example #4 - write the formula for diphosphorous pentoxide.
Step #1 - the first symbol is P and the subscript is 2.
Step #2 - pentoxide says five oxygens are involved.
The formula of this compound is P2O5.
Example #5 - write the formula for iodine heptafluoride.
Step #1 - the first symbol is I and the subscript is 1. Again it is understood to be there.
Step #2 - heptafluoride says 7 florides are involved.
The formula of this compound is IF7.
| given name, write formula (practice) | |
| File Size: | 16 kb |
| File Type: | docx |
These compounds to follow ARE NOT binary compounds. They contain three or more elements, as opposed to only two in a binary compound.
The Greek method WILL NOT be used. That naming technique is used only for binary compounds of two nonmetals. That means, if you see a formula like BaSO4, the name is not barium monosulfur tetraoxide. Another example: Na2SO3 is not named disodium monosulfur trioxide.
Consequently, a warning: it is important that you learn to recognize the presence of a polyatomic ion in a formula.
The cations used will be a mix of fixed charges AND variable charges. You must know which are which.
Another warning: you must also know the charges associated with each polyatomic ion. For example, NO3¯ is called nitrate and it has a minus one charge. Many people have thought this means nitrate has a minus three charge. IT DOES NOT. Another example along these lines is NH4+, the ammonium ion. It DOES NOT have a +4 charge, it has a +1 charge.
Use of Parenthesis When more than one polyatomic ion is required, parenthesis are used to enclose the ion with the subscript going outside the parenthesis. For example, the very first formula used is Fe(NO3)2. This means that two NO3¯ are involved in the compound. Without the parenthesis, the formula would be FeNO32, a far cry from the correct formula.
When you say a formula involving parenthesis out loud, you use the word "taken" as in the formula for ammonium sulfide, which is (NH4)2S. Out loud, you say "N H four taken twice S." OR with the formula for copper(II) chlorate, which is Cu(ClO3)2. You say " Cu Cl O three taken twice."
Example #1 - write the name for Fe(NO3)2 Step #1 - decide if the cation is one showing variable charge. If so, a Roman numeral will be needed. In this case, iron does show variable charge.
If a variable charge cation is involved, you must determine the Roman numeral involved. You do this by computing the total charge contributed by the polyatomic ion. In this case, NO3¯ has a minus one charge and there are two of them, making a total of minus 2.
Therefore, the iron must be a positive two, in order to keep the total charge of the formula at zero.
Step #2 - determine the name of the polyatomic ion. Nitrate is the name of NO3¯.
The correct name is iron(II) nitrate. The common name would be ferrous nitrate.
Example #2 - write the name for NaOH
Step #1 - the cation, Na+, does not show a variable charge, so no Roman numeral is needed. The name is sodium.
Step#2 - OH¯ is recognized as the hydroxide ion.
The name of this compound is sodium hydroxide.
Example #3 - write the name for KMnO4
Step #1 - the cation, K+, does not show a variable charge, so no Roman numeral is needed. The name is potassium.
Step#2 - MnO4¯ is recognized as the permanganate ion.
The name of this compound is potassium permanganate.
Example #4 - write the name for Cu2SO4 Step #1 - decide if the cation is one showing variable charge. If so, a Roman numeral will be needed. In this case, copper does show variable charge.
If a variable charge cation is involved, you must determine the Roman numeral involved. You do this by computing the total charge contributed by the polyatomic ion. In this case, SO42¯ has a minus two charge and there is only one, making a total of minus 2.
Therefore, the copper must be a positive one. Why? Well, there must be a positive two to go with the negative two in order to make zero. Since the formula shows two copper atoms involved, each must be a plus one charge.
Step #2 - determine the name of the polyatomic ion. Sulfate is the name of SO42¯.
The correct name is copper(I) sulfate. The common name would be cuprous sulfate.
Example #5 - write the name for Ca(ClO3)2
The first part of the name comes from the first element's name: calcium. You also determine that it is not a cation of variable charge.
The second part of the name comes from the name of the polyatomic ion: chlorate.
This compound is named calcium chlorate.
Example #6 - write the name for Fe(OH)3
Iron is an element with two possible oxidation states. The iron is a +3 charge because (1) there are three hydroxides, (2) hydroxide is a minus one charge, (3) this gives a total charge of negative three and (40 there is only one iron, so it must be a +3.
Therefore the first part of the name is iron(III).
The second part of the name is hydroxide, the name of the polyatomic ion.
The name of this compound is iron(III) hydroxide (or ferric hydroxide when using the common method).
The Greek method WILL NOT be used. That naming technique is used only for binary compounds of two nonmetals. That means, if you see a formula like BaSO4, the name is not barium monosulfur tetraoxide. Another example: Na2SO3 is not named disodium monosulfur trioxide.
Consequently, a warning: it is important that you learn to recognize the presence of a polyatomic ion in a formula.
The cations used will be a mix of fixed charges AND variable charges. You must know which are which.
Another warning: you must also know the charges associated with each polyatomic ion. For example, NO3¯ is called nitrate and it has a minus one charge. Many people have thought this means nitrate has a minus three charge. IT DOES NOT. Another example along these lines is NH4+, the ammonium ion. It DOES NOT have a +4 charge, it has a +1 charge.
Use of Parenthesis When more than one polyatomic ion is required, parenthesis are used to enclose the ion with the subscript going outside the parenthesis. For example, the very first formula used is Fe(NO3)2. This means that two NO3¯ are involved in the compound. Without the parenthesis, the formula would be FeNO32, a far cry from the correct formula.
When you say a formula involving parenthesis out loud, you use the word "taken" as in the formula for ammonium sulfide, which is (NH4)2S. Out loud, you say "N H four taken twice S." OR with the formula for copper(II) chlorate, which is Cu(ClO3)2. You say " Cu Cl O three taken twice."
Example #1 - write the name for Fe(NO3)2 Step #1 - decide if the cation is one showing variable charge. If so, a Roman numeral will be needed. In this case, iron does show variable charge.
If a variable charge cation is involved, you must determine the Roman numeral involved. You do this by computing the total charge contributed by the polyatomic ion. In this case, NO3¯ has a minus one charge and there are two of them, making a total of minus 2.
Therefore, the iron must be a positive two, in order to keep the total charge of the formula at zero.
Step #2 - determine the name of the polyatomic ion. Nitrate is the name of NO3¯.
The correct name is iron(II) nitrate. The common name would be ferrous nitrate.
Example #2 - write the name for NaOH
Step #1 - the cation, Na+, does not show a variable charge, so no Roman numeral is needed. The name is sodium.
Step#2 - OH¯ is recognized as the hydroxide ion.
The name of this compound is sodium hydroxide.
Example #3 - write the name for KMnO4
Step #1 - the cation, K+, does not show a variable charge, so no Roman numeral is needed. The name is potassium.
Step#2 - MnO4¯ is recognized as the permanganate ion.
The name of this compound is potassium permanganate.
Example #4 - write the name for Cu2SO4 Step #1 - decide if the cation is one showing variable charge. If so, a Roman numeral will be needed. In this case, copper does show variable charge.
If a variable charge cation is involved, you must determine the Roman numeral involved. You do this by computing the total charge contributed by the polyatomic ion. In this case, SO42¯ has a minus two charge and there is only one, making a total of minus 2.
Therefore, the copper must be a positive one. Why? Well, there must be a positive two to go with the negative two in order to make zero. Since the formula shows two copper atoms involved, each must be a plus one charge.
Step #2 - determine the name of the polyatomic ion. Sulfate is the name of SO42¯.
The correct name is copper(I) sulfate. The common name would be cuprous sulfate.
Example #5 - write the name for Ca(ClO3)2
The first part of the name comes from the first element's name: calcium. You also determine that it is not a cation of variable charge.
The second part of the name comes from the name of the polyatomic ion: chlorate.
This compound is named calcium chlorate.
Example #6 - write the name for Fe(OH)3
Iron is an element with two possible oxidation states. The iron is a +3 charge because (1) there are three hydroxides, (2) hydroxide is a minus one charge, (3) this gives a total charge of negative three and (40 there is only one iron, so it must be a +3.
Therefore the first part of the name is iron(III).
The second part of the name is hydroxide, the name of the polyatomic ion.
The name of this compound is iron(III) hydroxide (or ferric hydroxide when using the common method).
| given formula, write name (practice) | |
| File Size: | 18 kb |
| File Type: | docx |
Another warning: you must also know the formula and charge associated with each polyatomic ion's name. For example, NO3¯ is called nitrate and it has a minus one charge. The formula and charge are not inherent in the name.
Use of Parenthesis When more than one polyatomic ion is required, parenthesis are used to enclose the ion with the subscript going outside the parenthesis. For example, the first example used below is copper(II) chlorate. The correct formula will require the use of parenthesis.
When you say a formula involving parenthesis out loud, you use the word "taken" as in the formula for ammonium sulfide, which is (NH4)2S. Out loud, you say "N H four taken twice S." OR with the formula for copper(II) nitrate, which is Cu(NO3)2. You say " Cu N O three taken twice."
On writing the formula from the ions, you may want to review Charge-Crossing.html or Least-Common-Multiple.html for more information.
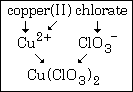
Example #1 - write the formula for copper(II) chlorate
Step #1 - the first word tells you the symbol of the cation. In this case it is Cu.
Step #2 - the Roman numeral WILL tell you the charge on the cation. In this case it is a positive two.
Step #3 - the polyatomic formula and charge comes from the second name. In this case, chlorate means ClO3¯.
Step #4 - remembering the rule that a formula must have zero total charge, you write the formula Cu(ClO3)2.
This graphic summarizes example #1:
Use of Parenthesis When more than one polyatomic ion is required, parenthesis are used to enclose the ion with the subscript going outside the parenthesis. For example, the first example used below is copper(II) chlorate. The correct formula will require the use of parenthesis.
When you say a formula involving parenthesis out loud, you use the word "taken" as in the formula for ammonium sulfide, which is (NH4)2S. Out loud, you say "N H four taken twice S." OR with the formula for copper(II) nitrate, which is Cu(NO3)2. You say " Cu N O three taken twice."
On writing the formula from the ions, you may want to review Charge-Crossing.html or Least-Common-Multiple.html for more information.
Example #1 - write the formula for copper(II) chlorate
Step #1 - the first word tells you the symbol of the cation. In this case it is Cu.
Step #2 - the Roman numeral WILL tell you the charge on the cation. In this case it is a positive two.
Step #3 - the polyatomic formula and charge comes from the second name. In this case, chlorate means ClO3¯.
Step #4 - remembering the rule that a formula must have zero total charge, you write the formula Cu(ClO3)2.
This graphic summarizes example #1:
By the way, out load, this is said C U C L O three taken twice.
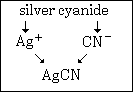
Example #2 - write the formula for silver cyanide
Step #1 - the first word tells you the symbol of the cation. In this case it is Ag+.
Step #2 - silver has a constant charge of +1, it is not a cation with variable charge.
Step #3 - the polyatomic formula and charge comes from the second name. In this case, cyanide means CN¯.
Step #4 - remembering the rule that a formula must have zero total charge, you write the formula AgCN.
This graphic summarizes example #2:
Example #2 - write the formula for silver cyanide
Step #1 - the first word tells you the symbol of the cation. In this case it is Ag+.
Step #2 - silver has a constant charge of +1, it is not a cation with variable charge.
Step #3 - the polyatomic formula and charge comes from the second name. In this case, cyanide means CN¯.
Step #4 - remembering the rule that a formula must have zero total charge, you write the formula AgCN.
This graphic summarizes example #2:
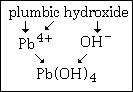
Example #3 - write the formula for plumbic hydroxide
Step #1 - the cation, Pb4+, does show a variable charge. The "-ic" ending means the higher of the two, for this cation that means +4.
Step#2 - hydroxide is recognized as OH¯.
The formula of this compound is Pb(OH)4. Notice that it is not PbOH4.
This graphic summarizes example #3:
Step #1 - the cation, Pb4+, does show a variable charge. The "-ic" ending means the higher of the two, for this cation that means +4.
Step#2 - hydroxide is recognized as OH¯.
The formula of this compound is Pb(OH)4. Notice that it is not PbOH4.
This graphic summarizes example #3:
Example #4 - write the formula for sodium phosphate
Step #1 - the cation, sodium, is Na+, and it does not show a variable charge.
Step#2 - phosphate is PO43¯.
The formula of this compound is Na3PO4. Notice that no parenthesis are required since only one polyatomis is used.
This graphic summarizes example #4:
Step #1 - the cation, sodium, is Na+, and it does not show a variable charge.
Step#2 - phosphate is PO43¯.
The formula of this compound is Na3PO4. Notice that no parenthesis are required since only one polyatomis is used.
This graphic summarizes example #4:
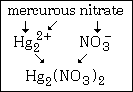
Example #5 - write the formula for mercurous nitrate
Step #1 - the cation, mercurous, does show a variable charge and its formula is unusual. It is Hg22+. The "-ous" ending indicates the lower of the two charges mercury shows and that is the +1 charge. Remember that, in this particular case, Hg+ is wrong.
Step#2 - nitrate is NO3¯.
The formula of this compound is Hg2(NO3)2. This formula is not reduced.
This graphic summarizes example #5:
Step #1 - the cation, mercurous, does show a variable charge and its formula is unusual. It is Hg22+. The "-ous" ending indicates the lower of the two charges mercury shows and that is the +1 charge. Remember that, in this particular case, Hg+ is wrong.
Step#2 - nitrate is NO3¯.
The formula of this compound is Hg2(NO3)2. This formula is not reduced.
This graphic summarizes example #5:
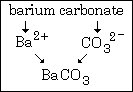
Example #6 - write the name for barium carbonate
Step #1 - the cation, barium, does not show a variable charge and its symbol is Ba2+.
Step#2 - carbonate is CO32¯.
The formula of this compound is BaCO3.
This graphic summarizes example #6:
Step #1 - the cation, barium, does not show a variable charge and its symbol is Ba2+.
Step#2 - carbonate is CO32¯.
The formula of this compound is BaCO3.
This graphic summarizes example #6:
Example #6 - write the name for mercury(I) phosphate
Step #1 - the cation, mercury(I), does show a variable charge and its symbol is Hg22+. Notice that there is a subscript of two and the charge is +2. Therefore, EACH Hg atom is +1, leading to the name mercury(I).
Step#2 - phosphate is PO43¯.
The formula of this compound is (Hg2)3(PO4)2.
Note the use of parenthesis around both parts (positive and negative) of the formula. It would be incorrect to write this formula: Hg6(PO4)2. We want the formula to show that mercury(I) comes in groups of two and that there are three of them.
Step #1 - the cation, mercury(I), does show a variable charge and its symbol is Hg22+. Notice that there is a subscript of two and the charge is +2. Therefore, EACH Hg atom is +1, leading to the name mercury(I).
Step#2 - phosphate is PO43¯.
The formula of this compound is (Hg2)3(PO4)2.
Note the use of parenthesis around both parts (positive and negative) of the formula. It would be incorrect to write this formula: Hg6(PO4)2. We want the formula to show that mercury(I) comes in groups of two and that there are three of them.
| given name, write formula (practice) | |
| File Size: | 17 kb |
| File Type: | docx |
Nomenclature of Inorganic Acids
Recognizing an Acid
At this very beginning level, you will recognize an acid by the fact that its formula starts with H, as in these examples:
HCl
HBr
HI
HNO3
HClO4
H2SO4
H3PO4
As you become more sophisticated in your chemistry, you will realize that there are many acid formulas that do not start with H, but those will almost all be left for another time.
There is one exception to this: the formula CH3COOH should be recognized as acetic acid. The particular way it is written is common in organic chemistry. An alternate way to write acetic acid is HC2H3O2.
One last comment before looking at how to name acids: the formula H2O should not be considered an acid. It is the formula for water. It is not an acid. When you study acid-base behavior later in the school year, you will learn more about water's role in acid-base chemistry, but not now.
Naming Acids
In order to explain acid naming, the sequence of HCl, HClO, HClO2, HClO3, and HClO4 will be discussed in order.
HCl is a binary acid. All binary acids are named the same way:
The name for HCl is hydrochloric acid. Other binary acids you are responsible for are HF, HBr, HI, and H2S.
1) HClO is an acid involving a polyatomic ion. You MUST recognize the polyatomic ion in the formula. There is no other way to figure out the name. If you don't recognize the polyatomic, then you're sunk without a trace.
The polyatomic ion is ClO¯ and its name is hypochlorite. Any time you see the "ite" suffix, you change it to "ous" and add the word acid.
The name of HClO is hypochlorous acid.
2) HClO2 has the ClO2¯ polyatomic ion in it. The name of this ion is chlorite.
Since the "ite" suffix is used, it gets changed to "ous."
The name of HClO2 is chlorous acid.
3) HClO3 has the ClO3¯ polyatomic ion and its name is chlorate. Any time you know the "ate" ending is used on the polyatomic, you use "ic" when you write the corresponding acid formula.
The name of HClO3 is chloric acid.
4) HClO4 has the ClO4¯ polyatomic ion and its name is perchlorate.
Since the "ate" suffix is used, it gets changed to "ic."
The name of HClO4 is perchloric acid.
You have to:
Recognizing an Acid
At this very beginning level, you will recognize an acid by the fact that its formula starts with H, as in these examples:
HCl
HBr
HI
HNO3
HClO4
H2SO4
H3PO4
As you become more sophisticated in your chemistry, you will realize that there are many acid formulas that do not start with H, but those will almost all be left for another time.
There is one exception to this: the formula CH3COOH should be recognized as acetic acid. The particular way it is written is common in organic chemistry. An alternate way to write acetic acid is HC2H3O2.
One last comment before looking at how to name acids: the formula H2O should not be considered an acid. It is the formula for water. It is not an acid. When you study acid-base behavior later in the school year, you will learn more about water's role in acid-base chemistry, but not now.
Naming Acids
In order to explain acid naming, the sequence of HCl, HClO, HClO2, HClO3, and HClO4 will be discussed in order.
HCl is a binary acid. All binary acids are named the same way:
- the prefix "hydro" is used.
- the root of the anion is used.
- the suffix "ic" is used.
- the word "acid" is used as the second word in the name.
The name for HCl is hydrochloric acid. Other binary acids you are responsible for are HF, HBr, HI, and H2S.
1) HClO is an acid involving a polyatomic ion. You MUST recognize the polyatomic ion in the formula. There is no other way to figure out the name. If you don't recognize the polyatomic, then you're sunk without a trace.
The polyatomic ion is ClO¯ and its name is hypochlorite. Any time you see the "ite" suffix, you change it to "ous" and add the word acid.
The name of HClO is hypochlorous acid.
2) HClO2 has the ClO2¯ polyatomic ion in it. The name of this ion is chlorite.
Since the "ite" suffix is used, it gets changed to "ous."
The name of HClO2 is chlorous acid.
3) HClO3 has the ClO3¯ polyatomic ion and its name is chlorate. Any time you know the "ate" ending is used on the polyatomic, you use "ic" when you write the corresponding acid formula.
The name of HClO3 is chloric acid.
4) HClO4 has the ClO4¯ polyatomic ion and its name is perchlorate.
Since the "ate" suffix is used, it gets changed to "ic."
The name of HClO4 is perchloric acid.
You have to:
- recognize when a polyatomic is present and
- know its name.
| acid nomenclature (practice) | |
| File Size: | 16 kb |
| File Type: | docx |